Abstract
Transition metal oxides with mobile electrons and oxygen ions (mixed conductors) constitute a broad class of materials that include selective
oxidation catalysts, high-temperature electrocatalysts, and ion-transport membrane materials. Although the thermodynamic and transport properties
of mixed conductors are generally understood, a consensus has not yet emerged regarding the mechanisms and rate laws governing exchange of oxygen
with the bulk at the gas-exposed surface. To aid interpretation of existing kinetic data, and generate testable hypotheses for further research,
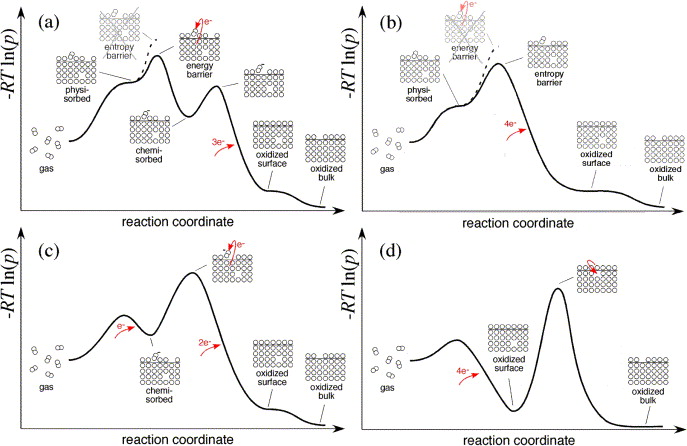
this paper outlines a framework for predicting O2 reduction rate laws based on specific reaction mechanisms. Based on nonequilibrium thermodynamics
and transition state theory, this framework yields rate laws that are rigorously consistent with thermodynamics, yet allow rates of individual steps
to be developed in terms of simple mass action laws, where appropriate. This framework is used to reexamine equilibrium oxygen-exchange kinetics
reported for electron-rich perovskite mixed conductors La1−xSrxCoO3−δ (LSC) and La1−xSrxFeO3−δ (LSF), which have a metallic and a semiconducting
band structure, respectively. Our analysis suggests that metallic band structure may play an important role in catalysis by stabilizing physisorbed
O2 on the surface. We also show that equilibrium surface exchange rates (as measured by 18O/16O isotopes, concentration steps, impedance, etc.)
are generally only weak indicators of mechanism, and emphasize the need for kinetic data involving moderate to large displacements from equilibrium.
Graphical Abstract